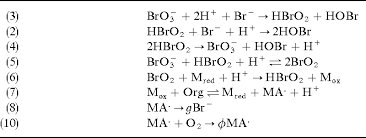Answer: The image depicts a series of chemical reactions, likely part of a mechanism for a complex reaction such as the Belousov-Zhabotinsky (BZ) reaction, which is a classic example of non-equilibrium thermodynamics.
Explanation: The reactions involve bromate (\( \text{BrO}_3^- \)), bromous acid (\( \text{HBrO}_2 \)), bromide (\( \text{Br}^- \)), and other species. These reactions likely illustrate the autocatalytic and oscillatory nature of the BZ reaction. Key concepts include redox reactions, radical formation, and catalytic cycles.
Steps:
- Reaction (3):
- Concepts: This is a redox reaction where bromate is reduced to bromous acid and hypobromous acid is formed.
- Reaction (2):
- Concepts: Further reduction of bromous acid and consumption of bromide ions.
- Reaction (4):
- Concepts: Disproportionation of bromous acid, regenerating bromate.
- Reaction (5):
- Concepts: Formation of bromine dioxide, an intermediate species.
- Reaction (6):
- Concepts: Redox reaction involving a metal catalyst.
- Reaction (7):
- Concepts: Organic substrate oxidation, regenerating the reduced metal catalyst.
- Reaction (8):
- Concepts: Decomposition of an intermediate, releasing bromide ions.
- Reaction (9):
- Concepts: Reaction with oxygen, possibly indicating radical formation or stabilization.
These reactions together illustrate the complex interplay of oxidation and reduction, radical formation, and catalytic cycles typical of oscillating chemical reactions.