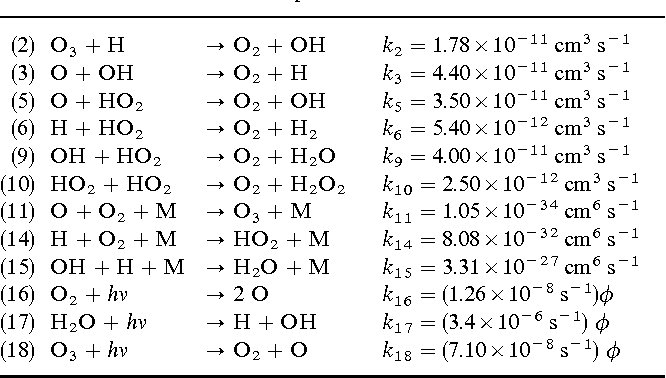
Answer: The image lists a series of chemical reactions with their corresponding rate constants and units. These reactions are part of atmospheric chemistry, specifically related to ozone and hydroxyl radical interactions.
Explanation: The table shows chemical reactions involving ozone (\(O_3\)), hydroxyl radicals (\(OH\)), and other species. Each reaction has a rate constant (\(k\)) with units indicating the reaction order. The reactions are part of a kinetic study, useful for understanding atmospheric processes like ozone depletion and formation.
Steps:
- Identify Reactions and Rate Constants:
- Each reaction is labeled with a number and involves reactants transforming into products.
- The rate constant (\(k\)) is given for each reaction, indicating the speed at which the reaction occurs.
- Units and Reaction Order:
- Units of \(cm^3 \, s^{-1}\) suggest bimolecular reactions (second-order).
- Units of \(s^{-1}\) suggest unimolecular reactions (first-order).
- Units involving \(\phi\) indicate photochemical reactions, where light (\(h\nu\)) is a reactant.
- Chemical Concepts:
- Photodissociation: Reactions (16), (17), and (18) involve photodissociation, where molecules absorb light and break into smaller species.
- Radical Reactions: Many reactions involve radicals (e.g., \(OH\), \(HO_2\)), which are highly reactive and crucial in atmospheric chemistry.
- Atmospheric Implications:
- These reactions are part of the ozone cycle, influencing ozone concentration in the stratosphere and troposphere.
- Understanding these reactions helps in modeling atmospheric phenomena and assessing environmental impacts.
This analysis provides insight into the chemical kinetics of atmospheric reactions, highlighting the importance of rate constants and reaction mechanisms in environmental chemistry.