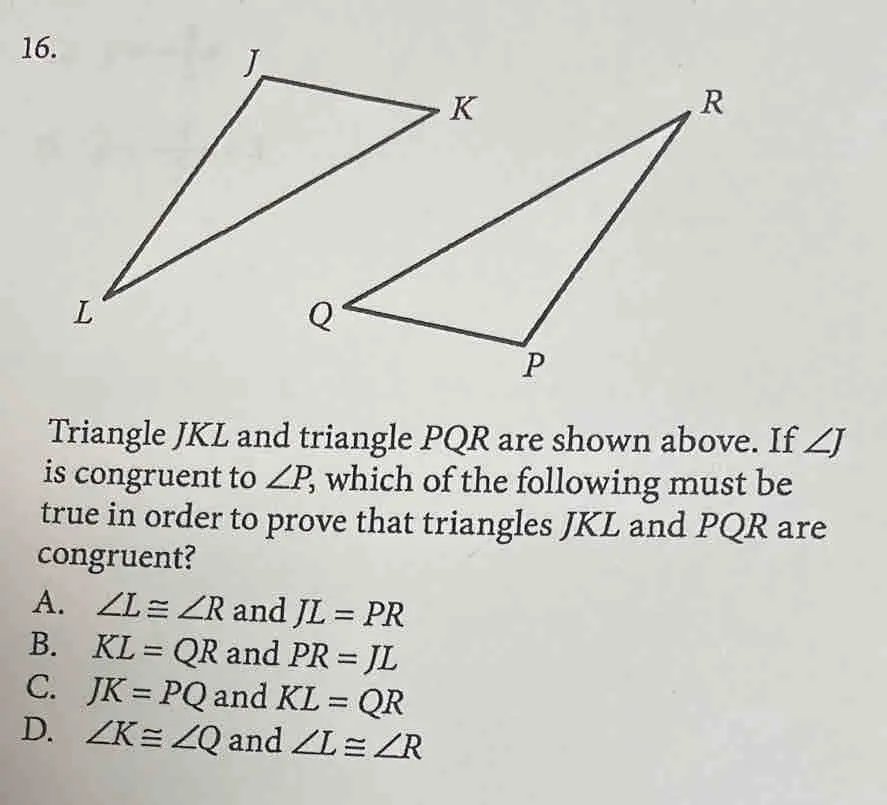
Answer: A. \(\angle L \equiv \angle R\) and \(JL = PR\)
Explanation: To prove that triangles \(JKL\) and \(PQR\) are congruent, we can use the Angle-Side-Angle (ASA) Congruence Theorem. This theorem states that if two angles and the included side of one triangle are congruent to two angles and the included side of another triangle, then the triangles are congruent.
Steps:
- Identify Given Information:
- \(\angle J \equiv \angle P\) is given.
- Apply ASA Congruence Theorem:
- We need another pair of congruent angles and the side between them to apply ASA.
- Option A provides \(\angle L \equiv \angle R\) and \(JL = PR\).
- With \(\angle J \equiv \angle P\), \(\angle L \equiv \angle R\), and the included side \(JL = PR\), the conditions for ASA are satisfied.
- Conclusion:
- By ASA, triangles \(JKL\) and \(PQR\) are congruent if \(\angle L \equiv \angle R\) and \(JL = PR\).
Thus, option A is the correct choice to prove the triangles are congruent using the ASA Congruence Theorem.