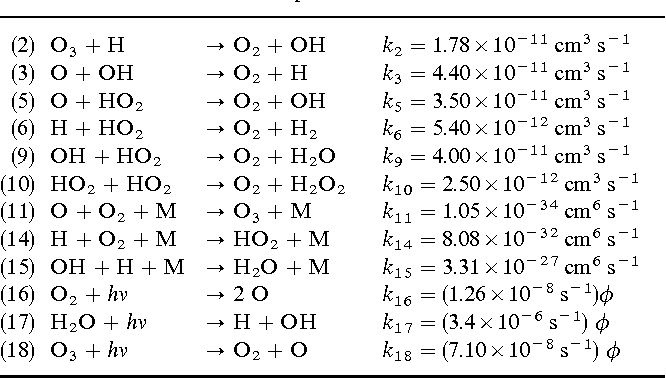
Answer: The image lists chemical reactions with their respective rate constants, which are used in atmospheric chemistry to model the behavior of ozone and related species.
Explanation: The table shows a series of chemical reactions involving ozone (\( \text{O}_3 \)), hydroxyl radicals (\( \text{OH} \)), and other species. Each reaction is associated with a rate constant (\( k \)), which indicates the speed of the reaction. These constants are essential for calculating reaction rates in atmospheric models. The units of the rate constants suggest the order of the reactions (e.g., bimolecular reactions have units of \( \text{cm}^3 \, \text{s}^{-1} \), while unimolecular reactions have units of \( \text{s}^{-1} \)).
Steps:
- Identify Reaction Types:
- Bimolecular Reactions: Involve two reactants. Examples include reactions (2), (3), (5), (6), (9), and (10). The rate constants for these reactions have units of \( \text{cm}^3 \, \text{s}^{-1} \).
- Termolecular Reactions: Involve three reactants, typically two molecules and a third body (M) to stabilize the reaction. Examples include reactions (11), (14), and (15). The rate constants have units of \( \text{cm}^6 \, \text{s}^{-1} \).
- Photodissociation Reactions: Involve the absorption of a photon (\( h\nu \)). Examples include reactions (16), (17), and (18). The rate constants are modified by the actinic flux (\( \phi \)).
- Use of Rate Constants:
- Rate Law: For a bimolecular reaction \( A + B \rightarrow \text{products} \), the rate is given by \( \text{Rate} = k[A][B] \).
- For Termolecular Reactions: The rate is \( \text{Rate} = k[A][B][M] \).
- For Photodissociation: The rate is \( \text{Rate} = k\phi \).
- Application in Atmospheric Chemistry:
- These reactions are part of the ozone cycle, which is crucial for understanding the formation and depletion of ozone in the atmosphere.
- The reactions involving \( \text{O}_3 \) and \( \text{OH} \) are particularly important for modeling the oxidative capacity of the atmosphere.
By analyzing these reactions and their rate constants, scientists can simulate and predict changes in atmospheric composition, which is vital for understanding environmental and climate-related processes.