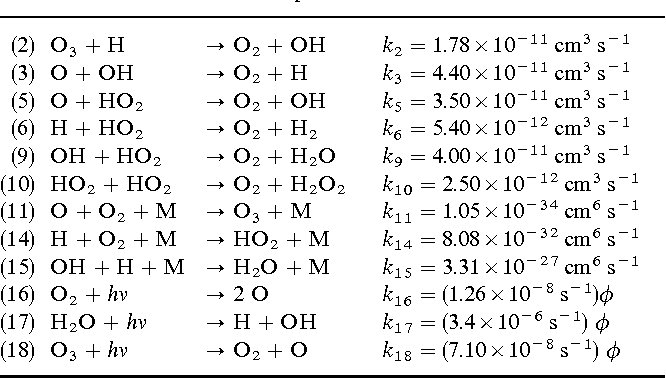
Answer: The image presents a series of chemical reactions with their respective rate constants, which are used to describe the kinetics of atmospheric reactions involving ozone (\(O_3\)), hydroxyl radicals (\(OH\)), and other species.
Explanation: The table lists various chemical reactions, each with a specific rate constant (\(k\)) and units. These reactions are part of atmospheric chemistry, particularly related to ozone depletion and formation. The rate constants indicate the speed of each reaction, which is crucial for modeling atmospheric processes.
Steps:
- Identify Reactions:
- Each line represents a chemical reaction with reactants on the left and products on the right.
- For example, reaction (2) is \(O_3 + H \rightarrow O_2 + OH\).
- Rate Constants:
- Each reaction has an associated rate constant (\(k\)), which quantifies the reaction rate.
- The units of \(k\) vary depending on the reaction order (e.g., \(cm^3 \, s^{-1}\) for bimolecular reactions).
- Photochemical Reactions:
- Reactions (16), (17), and (18) involve photodissociation, indicated by \(h\nu\), representing the energy from light.
- The rate constants for these reactions are modified by the photolysis rate (\(\phi\)).
- Atmospheric Implications:
- These reactions are part of the Chapman cycle and other processes that describe ozone dynamics in the stratosphere.
- Understanding these reactions helps in modeling the effects of pollutants and natural processes on ozone concentration.
- Mathematical Concepts:
- Rate Laws: The rate of a reaction is proportional to the product of the concentrations of the reactants, each raised to a power corresponding to its stoichiometric coefficient.
- Reaction Order: Determined by the sum of the powers of the concentration terms in the rate law.
By analyzing these reactions and their rate constants, scientists can predict changes in atmospheric composition and assess the impact of human activities on ozone levels.