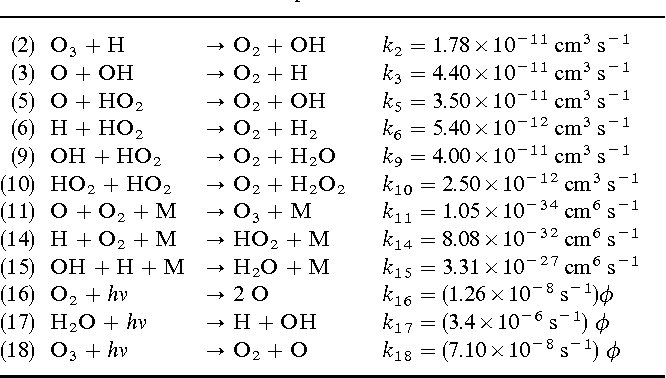
Answer: The image contains a list of chemical reactions with their respective rate constants and units. These reactions are part of atmospheric chemistry, specifically involving ozone (O₃) and hydroxyl radicals (OH).
Explanation: The table provides a set of chemical reactions with associated rate constants \( k \), which are crucial for understanding reaction kinetics in atmospheric chemistry. The reactions involve ozone (O₃), hydroxyl radicals (OH), and other species, indicating processes like ozone decomposition and radical formation. The units of the rate constants suggest whether the reactions are bimolecular (cm³ s⁻¹) or unimolecular (s⁻¹).
Steps:
- Identify Reaction Types:
- Bimolecular reactions: Involve two reactants and have rate constants with units of cm³ s⁻¹.
- Unimolecular reactions: Involve one reactant decomposing or reacting with light (photolysis) and have rate constants with units of s⁻¹.
- Analyze Each Reaction:
- Reaction (2): \( \text{O}_3 + \text{H} \rightarrow \text{O}_2 + \text{OH} \)
- Bimolecular reaction with \( k_2 = 1.78 \times 10^{-11} \, \text{cm}^3 \, \text{s}^{-1} \).
- Reaction (3): \( \text{O} + \text{OH} \rightarrow \text{O}_2 + \text{H} \)
- Bimolecular reaction with \( k_3 = 4.40 \times 10^{-11} \, \text{cm}^3 \, \text{s}^{-1} \).
- Reaction (5): \( \text{O} + \text{HO}_2 \rightarrow \text{O}_2 + \text{OH} \)
- Bimolecular reaction with \( k_5 = 3.50 \times 10^{-11} \, \text{cm}^3 \, \text{s}^{-1} \).
- Reaction (6): \( \text{H}_2 + \text{O} \rightarrow \text{OH} + \text{H} \)
- Bimolecular reaction with \( k_6 = 5.40 \times 10^{-11} \, \text{cm}^3 \, \text{s}^{-1} \).
- Reaction (9): \( \text{OH} + \text{HO}_2 \rightarrow \text{O}_2 + \text{H}_2\text{O} \)
- Bimolecular reaction with \( k_9 = 4.00 \times 10^{-11} \, \text{cm}^3 \, \text{s}^{-1} \).
- Reaction (10): \( \text{HO}_2 + \text{HO}_2 \rightarrow \text{O}_2 + \text{H}_2\text{O}_2 \)
- Bimolecular reaction with \( k_{10} = 2.50 \times 10^{-12} \, \text{cm}^3 \, \text{s}^{-1} \).
- Reaction (11): \( \text{O}_3 + \text{O} + \text{M} \rightarrow \text{O}_3 + \text{M} \)
- Termolecular reaction with \( k_{11} = 1.05 \times 10^{-34} \, \text{cm}^6 \, \text{s}^{-1} \).
- Reaction (14): \( \text{H} + \text{O}_2 + \text{M} \rightarrow \text{HO}_2 + \text{M} \)
- Termolecular reaction with \( k_{14} = 8.08 \times 10^{-32} \, \text{cm}^6 \, \text{s}^{-1} \).
- Reaction (15): \( \text{OH} + \text{H} + \text{M} \rightarrow \text{H}_2\text{O} + \text{M} \)
- Termolecular reaction with \( k_{15} = 3.31 \times 10^{-27} \, \text{cm}^6 \, \text{s}^{-1} \).
- Reaction (16): \( \text{O}_2 + h\nu \rightarrow 2\text{O} \)
- Photolysis with \( k_{16} = (1.26 \times 10^{-8} \, \text{s}^{-1})\phi \).
- Reaction (17): \( \text{H}_2\text{O} + h\nu \rightarrow \text{H} + \text{OH} \)
- Photolysis with \( k_{17} = (3.4 \times 10^{-6} \, \text{s}^{-1})\phi \).
- Reaction (18): \( \text{O}_3 + h\nu \rightarrow \text{O}_2 + \text{O} \)
- Photolysis with \( k_{18} = (7.10 \times 10^{-8} \, \text{s}^{-1})\phi \).
- Concepts Involved:
- Reaction Kinetics: Understanding how reaction rates are affected by concentration and the presence of a third body (M) in termolecular reactions.
- Photolysis: The decomposition of molecules by photons, crucial in atmospheric reactions involving sunlight.
- Atmospheric Chemistry: The study of chemical processes in the Earth’s atmosphere, particularly involving ozone and radicals like OH and HO₂.