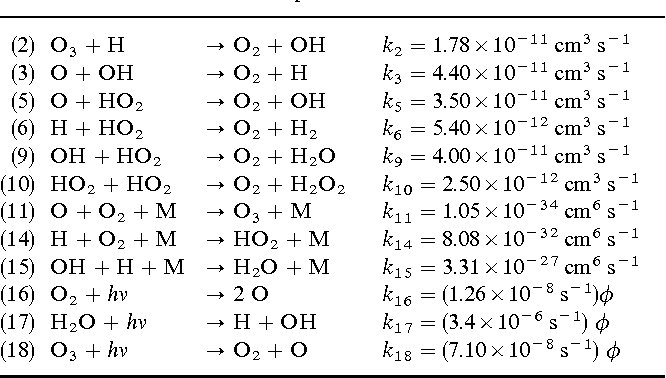
Answer: The image lists chemical reactions with their respective rate constants, indicating the speed of each reaction. It involves concepts from chemical kinetics, such as reaction rates and rate constants.
Explanation: The table provides a list of chemical reactions along with their rate constants, denoted by \( k \). These constants are crucial in determining the speed of each reaction and are expressed in units of \( \text{cm}^3/\text{s} \) or \( \text{s}^{-1} \), depending on the reaction order. The reactions involve species like ozone (\( \text{O}_3 \)), hydroxyl radicals (\( \text{OH} \)), and others, indicating a focus on atmospheric chemistry.
Steps:
- Identify Reaction Types:
- Each reaction involves different atmospheric species interacting, such as ozone (\( \text{O}_3 \)), hydroxyl radicals (\( \text{OH} \)), and hydrogen peroxide (\( \text{H}_2\text{O}_2 \)).
- Determine Reaction Order:
- The units of the rate constants provide clues about the reaction order:
- \( \text{cm}^3/\text{s} \) suggests a bimolecular reaction (second order).
- \( \text{s}^{-1} \) suggests a unimolecular reaction (first order).
- Analyze Photochemical Reactions:
- Reactions (16), (17), and (18) involve \( h\nu \), indicating photolysis reactions where light energy (\( h\nu \)) breaks chemical bonds.
- Use of Rate Constants:
- The rate of a reaction can be calculated using the rate constant and the concentrations of the reactants. For a bimolecular reaction, the rate is given by:
- For a unimolecular reaction:
- Application in Atmospheric Chemistry:
- These reactions are part of the ozone cycle and other atmospheric processes, crucial for understanding phenomena like ozone depletion and formation of secondary pollutants.
By analyzing the table, one can understand the dynamics of atmospheric reactions and their implications for environmental chemistry.