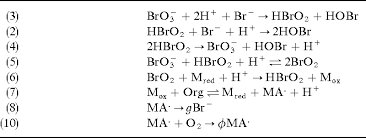Answer: The image shows a series of chemical reactions, likely part of a mechanism for a complex chemical process, such as an oscillating reaction like the Belousov-Zhabotinsky (BZ) reaction.
Explanation: This sequence of reactions involves bromate (\( \text{BrO}_3^- \)), bromous acid (\( \text{HBrO}_2 \)), and other bromine species, which are typical in the BZ reaction. The reactions demonstrate redox processes, radical formation, and catalytic cycles.
Steps:
- Reaction (3):
- Concept: Redox reaction.
- Description: Bromate (\( \text{BrO}_3^- \)) is reduced to bromous acid (\( \text{HBrO}_2 \)), while bromide (\( \text{Br}^- \)) is oxidized to hypobromous acid (\( \text{HOBr} \)).
- Reaction (2):
- Concept: Acid-base reaction.
- Description: Bromous acid (\( \text{HBrO}_2 \)) reacts with bromide (\( \text{Br}^- \)) and protons (\( \text{H}^+ \)) to form hypobromous acid (\( \text{HOBr} \)).
- Reaction (4):
- Concept: Radical formation.
- Description: Two molecules of bromous acid (\( \text{HBrO}_2 \)) react to form bromate (\( \text{BrO}_3^- \)), hypobromous acid (\( \text{HOBr} \)), and protons (\( \text{H}^+ \)).
- Reaction (5):
- Concept: Redox reaction.
- Description: Bromate (\( \text{BrO}_3^- \)) reacts with bromous acid (\( \text{HBrO}_2 \)) and protons (\( \text{H}^+ \)) to produce bromine dioxide (\( \text{BrO}_2 \)).
- Reaction (6):
- Concept: Redox reaction.
- Description: Bromine dioxide (\( \text{BrO}_2 \)) reacts with a reducing agent (\( \text{M}_{\text{red}} \)) and protons (\( \text{H}^+ \)) to regenerate bromous acid (\( \text{HBrO}_2 \)) and oxidized form of the reducing agent (\( \text{M}_{\text{ox}} \)).
- Reaction (7):
- Concept: Organic reaction.
- Description: An oxidizing agent (\( \text{M}_{\text{ox}} \)) reacts with an organic substrate (\( \text{Org} \)) to produce a reduced form (\( \text{M}_{\text{red}} \)) and a radical (\( \text{MA}^\cdot \)).
- Reaction (8):
- Concept: Radical reaction.
- Description: The radical (\( \text{MA}^\cdot \)) reacts to form a brominated product (\( \text{gBr} \)).
- Reaction (9):
- Concept: Radical reaction.
- Description: The radical (\( \text{MA}^\cdot \)) reacts with oxygen (\( \text{O}_2 \)) to form another radical (\( \phi \text{MA}^\cdot \)).
These reactions collectively illustrate the complex interplay of redox and radical processes typical in oscillating reactions, where intermediates are continuously regenerated, leading to periodic changes in concentration.