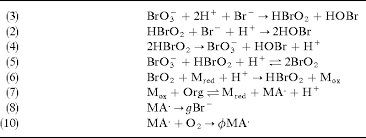Answer: The image depicts a series of chemical reactions, likely part of a mechanism, possibly related to a bromate oscillating reaction or a similar redox process.
Explanation: The reactions involve bromate (\( \text{BrO}_3^- \)), bromous acid (\( \text{HBrO}_2 \)), and bromine species, indicating a redox reaction sequence. The reactions also involve organic species and radicals, suggesting a complex mechanism possibly related to oscillating chemical reactions like the Belousov-Zhabotinsky reaction.
Steps:
- Reaction (3):
- \( \text{BrO}_3^- + 2\text{H}^+ + \text{Br}^- \rightarrow \text{HBrO}_2 + \text{HOBr} \)
- This is a redox reaction where bromate is reduced to bromous acid and hypobromous acid.
- Reaction (4):
- \( \text{HBrO}_2 + \text{Br}^- + \text{H}^+ \rightarrow 2\text{HOBr} \)
- Further reduction of bromous acid to hypobromous acid.
- Reaction (5):
- \( 2\text{HBrO}_2 \rightarrow \text{BrO}_3^- + \text{HOBr} + \text{H}^+ \)
- Disproportionation of bromous acid back to bromate and hypobromous acid.
- Reaction (6):
- \( \text{BrO}_2 + \text{M}_{\text{red}} + \text{H}^+ \rightarrow \text{HBrO}_2 + \text{M}_{\text{ox}} \)
- Redox reaction involving a mediator species (\( \text{M} \)).
- Reaction (7):
- \( \text{M}_{\text{ox}} + \text{Org} \rightarrow \text{M}_{\text{red}} + \text{MA}^+ + \text{H}^+ \)
- Oxidation of an organic species by the oxidized mediator.
- Reaction (8):
- \( \text{MA}^+ \rightarrow g\text{Br}^- \)
- Decomposition or transformation of an intermediate species.
- Reaction (9):
- \( \text{MA}^+ + \text{O}_2 \rightarrow \phi\text{MA}^+ \)
- Reaction involving oxygen, possibly indicating radical formation or propagation.
These reactions collectively suggest a complex mechanism involving multiple redox steps, radical intermediates, and possibly oscillatory behavior. The presence of organic species and radicals is typical in such systems, where the interplay of reactions leads to dynamic behavior.