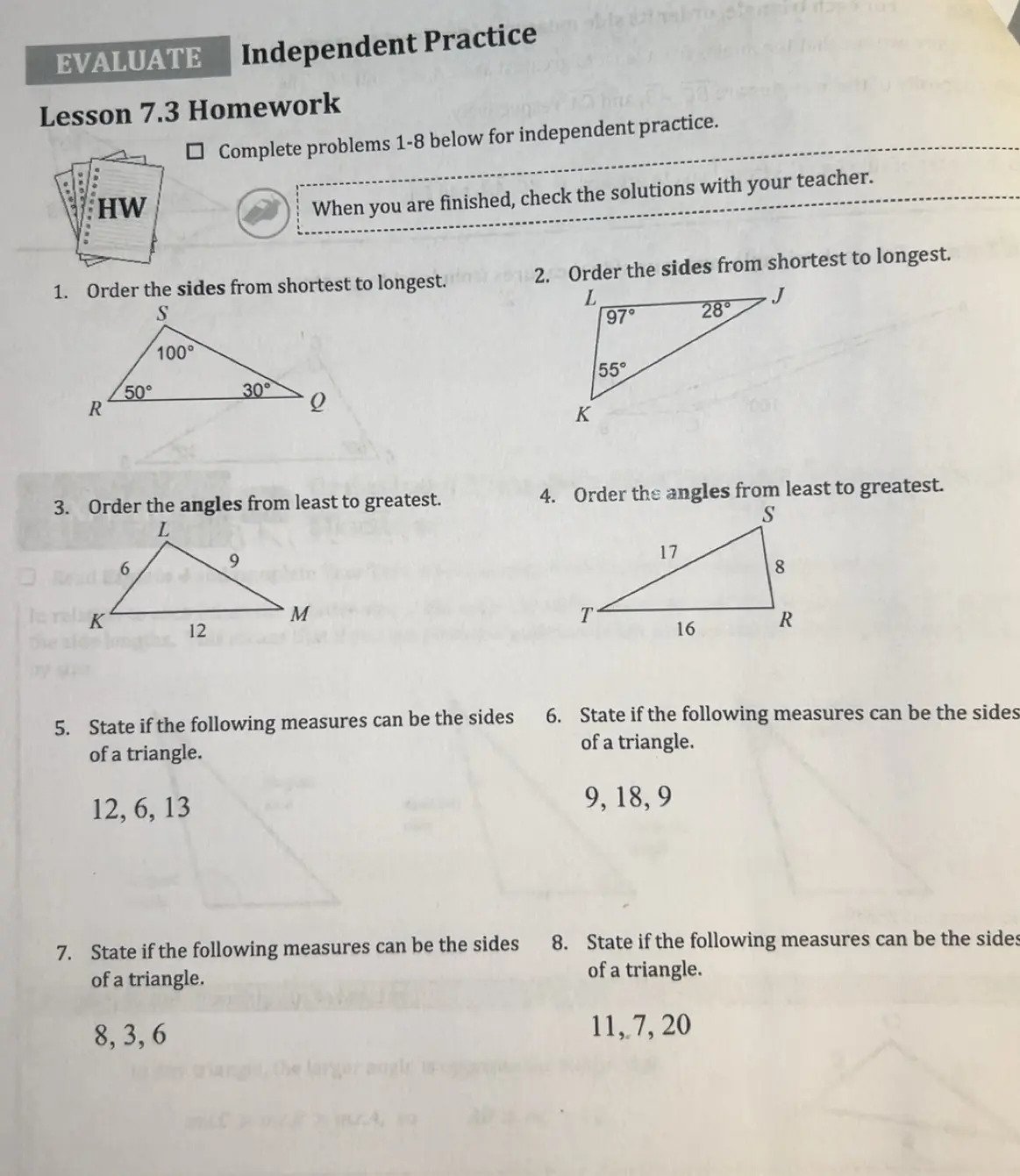
Answer:
- \( \overline{RQ}, \overline{SQ}, \overline{RS} \)
- \( \overline{KJ}, \overline{KL}, \overline{LJ} \)
- \( \angle K, \angle L, \angle M \)
- \( \angle R, \angle S, \angle T \)
- Yes
- No
- No
- No
Explanation:
- Triangle Inequality Theorem: The sides opposite the smallest angles are the shortest.
- Triangle Inequality Theorem: The sides opposite the smallest angles are the shortest.
- Triangle Inequality Theorem: The smallest angle is opposite the shortest side.
- Triangle Inequality Theorem: The smallest angle is opposite the shortest side.
5-8. Triangle Inequality Theorem: The sum of the lengths of any two sides must be greater than the length of the third side.
Steps:
- Order the sides from shortest to longest:
- Given angles: \( \angle R = 50^\circ \), \( \angle S = 100^\circ \), \( \angle Q = 30^\circ \).
- Shortest side is opposite the smallest angle: \( \overline{RQ} \).
- Next is \( \overline{SQ} \) opposite \( \angle R \).
- Longest is \( \overline{RS} \) opposite \( \angle S \).
- Order the sides from shortest to longest:
- Given angles: \( \angle L = 97^\circ \), \( \angle J = 28^\circ \), \( \angle K = 55^\circ \).
- Shortest side is opposite the smallest angle: \( \overline{KJ} \).
- Next is \( \overline{KL} \) opposite \( \angle K \).
- Longest is \( \overline{LJ} \) opposite \( \angle L \).
- Order the angles from least to greatest:
- Given sides: \( \overline{KL} = 6 \), \( \overline{LM} = 9 \), \( \overline{KM} = 12 \).
- Smallest angle is opposite the shortest side: \( \angle K \).
- Next is \( \angle L \) opposite \( \overline{LM} \).
- Largest is \( \angle M \) opposite \( \overline{KM} \).
- Order the angles from least to greatest:
- Given sides: \( \overline{TR} = 8 \), \( \overline{RS} = 17 \), \( \overline{TS} = 16 \).
- Smallest angle is opposite the shortest side: \( \angle R \).
- Next is \( \angle S \) opposite \( \overline{TS} \).
- Largest is \( \angle T \) opposite \( \overline{RS} \).
- State if the measures can be the sides of a triangle:
- Sides: 12, 6, 13.
- Check: \( 12 + 6 > 13 \), \( 12 + 13 > 6 \), \( 6 + 13 > 12 \).
- All conditions satisfied: Yes.
- State if the measures can be the sides of a triangle:
- Sides: 9, 18, 9.
- Check: \( 9 + 9 > 18 \) (False).
- Condition not satisfied: No.
- State if the measures can be the sides of a triangle:
- Sides: 8, 3, 6.
- Check: \( 8 + 3 > 6 \), \( 8 + 6 > 3 \), \( 3 + 6 > 8 \) (False).
- Condition not satisfied: No.
- State if the measures can be the sides of a triangle:
- Sides: 11, 7, 20.
- Check: \( 11 + 7 > 20 \) (False).
- Condition not satisfied: No.