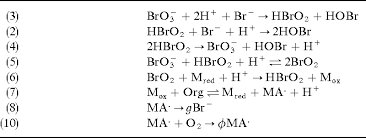Answer: The image depicts a series of chemical reactions, likely part of a mechanism for a complex chemical process such as a redox reaction or a catalytic cycle.
Explanation: The reactions involve bromate (\( \text{BrO}_3^- \)), bromous acid (\( \text{HBrO}_2 \)), and other bromine species, indicating a potential oscillating reaction like the Belousov-Zhabotinsky (BZ) reaction. This type of reaction is known for its periodic color changes and is a classic example of non-equilibrium thermodynamics in chemistry.
Steps:
- Reaction (3):
- This is a redox reaction where bromate is reduced to bromous acid.
- Reaction (2):
- Bromous acid reacts with bromide ions to form hypobromous acid.
- Reaction (4):
- This reaction shows the disproportionation of bromous acid.
- Reaction (5):
- A further redox reaction involving bromate and bromous acid.
- Reaction (6):
- Involves a redox reaction between bromine dioxide and a reducing agent.
- Reaction (7):
- An organic substrate is oxidized.
- Reaction (8):
- Possibly a decomposition or further reaction of an intermediate.
- Reaction (9):
- Involves interaction with oxygen, possibly indicating an oxidation step.
Key Concepts:
- Redox Reactions: Involves the transfer of electrons between species, changing their oxidation states.
- Oscillating Reactions: Characterized by periodic changes in concentration of reactants/products.
- Disproportionation: A specific type of redox reaction where a single substance is simultaneously oxidized and reduced.
This sequence of reactions highlights the complexity and interconnectivity of chemical processes, particularly in systems that exhibit non-linear dynamics like the BZ reaction.