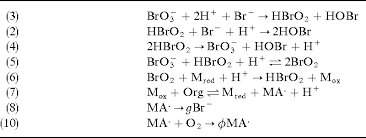Answer: The image contains a series of chemical reactions, not a mathematical problem. These reactions are part of a chemical mechanism, likely related to a redox process involving bromine species and organic compounds.
Explanation: The reactions listed are part of a chemical mechanism, possibly describing a catalytic cycle or a series of steps in a complex reaction. The reactions involve bromate (\( \text{BrO}_3^- \)), bromous acid (\( \text{HBrO}_2 \)), and other bromine species, as well as organic compounds and redox processes. The reactions suggest a sequence where bromine species are reduced and oxidized, and organic compounds are involved in redox transformations.
Steps:
- Reaction (3):
- \( \text{BrO}_3^- + 2\text{H}^+ + \text{Br}^- \rightarrow \text{HBrO}_2 + \text{HOBr} \)
- This is a reduction of bromate to bromous acid and hypobromous acid.
- Reaction (2):
- \( \text{HBrO}_2 + \text{Br}^- + \text{H}^+ \rightarrow 2\text{HOBr} \)
- Further reduction of bromous acid to hypobromous acid.
- Reaction (4):
- \( 2\text{HBrO}_2 \rightarrow \text{BrO}_3^- + \text{HOBr} + \text{H}^+ \)
- Disproportionation of bromous acid.
- Reaction (5):
- \( \text{BrO}_3^- + \text{HBrO}_2 + \text{H}^+ \rightarrow 2\text{BrO}_2 \)
- Formation of bromine dioxide.
- Reaction (6):
- \( \text{BrO}_2 + \text{M}_{\text{red}} + \text{H}^+ \rightarrow \text{HBrO}_2 + \text{M}_{\text{ox}} \)
- Redox reaction involving a metal or mediator.
- Reaction (7):
- \( \text{M}_{\text{ox}} + \text{Org} \rightarrow \text{M}_{\text{red}} + \text{MA}^+ + \text{H}^+ \)
- Oxidation of an organic compound, reducing the mediator.
- Reaction (8):
- \( \text{MA}^+ \rightarrow g\text{Br}^- \)
- Decomposition or transformation of an intermediate.
- Reaction (9):
- \( \text{MA}^+ + \text{O}_2 \rightarrow \phi\text{MA}^+ \)
- Reaction with oxygen, possibly regenerating or transforming the intermediate.
These reactions involve concepts such as redox chemistry, reaction mechanisms, and possibly catalytic cycles.