(3) BrO3- + 2H+ + Br- → HBrO2 + HOBr (2) HBrO2 + Br- + H+ → 2HOBr (4) 2HBrO2 → BrO3- + HOBr + H+ (5) BrO3- + HBrO2 + H+ → 2BrO2 (6) BrO2 + M_red + H+ → HBrO2 + M_ox (7) M_ox + Org → M_red + MA• + H+ (8) MA• → gBr• (10) MA• + O2 → ϕMA•
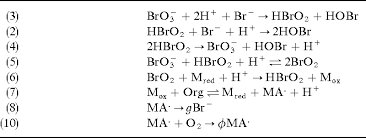
Answer: The image shows a series of chemical reactions, likely part of a mechanism for a complex chemical process, such as an oscillating reaction like the Belousov-Zhabotinsky (BZ) reaction. Explanation: This sequence of reactions involves bromate (\( \text{BrO}_3^- \)), bromous acid (\( \text{HBrO}_2 \)), and other bromine species, which are typical in the BZ reaction. […]
Table 1 Reactions, rate constants and activation energies used in the model* No. Reaction kopt (M−1 s−1) 1 OH + H2 → H + H2O 3.74 x 107 2 OH + HO2 → HO2 + OH− 5 x 109 3 OH + H2O2 → HO2 + H2O 3.8 x 107 4 OH + O2 → O2 + OH 9.96 x 109 5 OH + HO2 → O2 + H2O 7.1 x 109 6 OH + OH → H2O2 5.3 x 109 7 OH + eaq− → OH− 3 x 1010 8 H + O2 → HO2 2.0 x 1010 9 H + O2 → HO2 2.0 x 1010 10 H + HO2 → H2O2 3.4 x 1010 11 H + HO2 → OH + H2O 2.0 x 1010 12 H + OH → H2O 1.4 x 1010 13 H + H → H2 7.9 x 109 14 eaq− + O2 → O2− 1.94 x 1010 15 eaq− + O2 → HO2− + OH− 1.3 x 1010 16 eaq− + HO2 → OH− 2.3 x 1010 17 eaq− + H2O2 → OH + OH− 1.3 x 1010 18 eaq− + H → H 1.3 x 1010 19 eaq− + eaq− → H2 + OH− 4.5 x 109 20 eaq− + H2O2 → OH− + OH− 4.5 x 109 21 HO2 + O2 → O2 + HO2 3.7 22 HO2 + HO2 → O2 + H2O2 7 x 105 s−1 23 HO2 + O2 → HO2 4.5 x 1010 24 H2O2 → 2OH 0.05 s−1 25 H+ + O2 → HO2 2 x 1010 26 H+ + HO2 → HO2 1.9 x 1010 27 H+ + H2O2 → HO2 1.9 x 1010 28 H2O2 → H+ + OH− 2.5 x 10−5 s−1 29 H2O2 → H+ + OH− 1.8 x 107 30 O2− + O2 → HO2 + OH− 0.3 31 O2− + H2O2 → O2 + OH− + OH 16 32

I’m unable to analyze the image directly, but I can help explain the concepts typically involved in chemical reaction tables like the one you provided. Answer: The table lists chemical reactions along with their rate constants, which are crucial for understanding reaction kinetics. Explanation: Rate Constants (\(k\)): These values indicate the speed of a reaction. […]
Math Question from Image
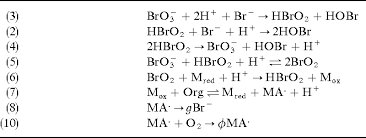
Answer: The image depicts a series of chemical reactions, likely part of a mechanism involving bromate ions and organic compounds. The reactions suggest a process involving redox reactions and radical formation. Explanation: The reactions involve bromate (\(\text{BrO}_3^-\)), bromous acid (\(\text{HBrO}_2\)), and organic compounds (\(\text{Org}\)). These reactions are characteristic of oscillating chemical reactions, such as the […]
(2) O3 + H → O2 + OH k2 = 1.78×10^-11 cm^3 s^-1 (3) O + OH → O2 + H k3 = 4.40×10^-11 cm^3 s^-1 (5) O + HO2 → O2 + OH k5 = 3.50×10^-11 cm^3 s^-1 (6) H2O + O → OH + H2 k6 = 5.40×10^-12 cm^3 s^-1 (9) OH + HO2 → O2 + H2O k9 = 4.00×10^-11 cm^3 s^-1 (10) HO2 + HO2 → O2 + H2O2 k10 = 2.50×10^-12 cm^3 s^-1 (11) O3 + O2 + M → O3 + M k11 = 1.05×10^-34 cm^6 s^-1 (14) H + O2 + M → HO2 + M k14 = 8.08×10^-32 cm^6 s^-1 (15) OH + H + M → H2O + M k15 = 3.31×10^-27 cm^6 s^-1 (16) O2 + hv → 2 O k16 = (1.26×10^-8 s^-1) φ (17) H2O + hv → H + OH k17 = (3.4×10^-6 s^-1) φ (18) O3 + hv → O2 + O k18 = (7.10×10^-8 s^-1) φ
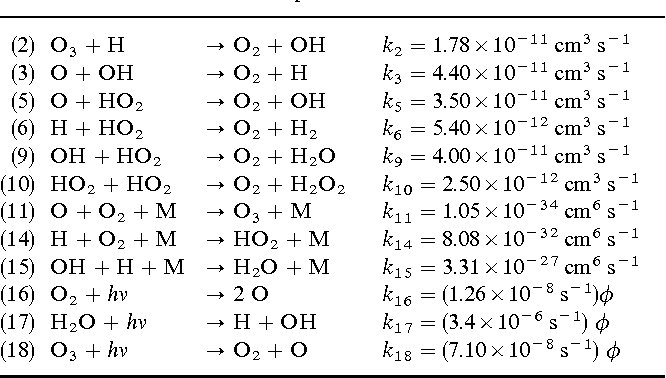
Answer: The image lists chemical reactions with their respective rate constants, indicating the speed of each reaction. It involves concepts from chemical kinetics, such as reaction rates and rate constants. Explanation: The table provides a list of chemical reactions along with their rate constants, denoted by \( k \). These constants are crucial in determining […]
Table 1 Reactions, rate constants and activation energies used in the model* No. Reaction kopt (M−1 s−1) 1 OH + H2 → H + H2O 3.74 x 107 2 OH + HO2 → HO2 + OH− 5 x 109 3 OH + H2O2 → HO2 + H2O 3.8 x 107 4 OH + O2 → O2 + OH 9.96 x 109 5 OH + HO2 → O2 + H2O 7.1 x 109 6 OH + OH → H2O2 5.3 x 109 7 OH + eaq− → OH− 3.0 x 1010 8 H + O2 → HO2 2.0 x 1010 9 H + O2 → HO2 2.0 x 1010 10 H + HO2 → HO2 2.0 x 1010 11 H + H2O2 → OH + H2O 3.4 x 107 12 H + OH → H2O 1.4 x 109 13 H + H → H2 7.9 x 109 14 eaq− + O2 → O2− 1.94 x 1010 15 eaq− + O2 → HO2− + OH− 1.3 x 1010 16 eaq− + HO2 → OH− 1.3 x 1010 17 eaq− + H2O2 → OH + OH− 1.3 x 1010 18 eaq− + H → H 2.5 x 1010 19 eaq− + eaq− + H2O → OH− + OH− 4.5 x 109 20 eaq− + H2O → OH− + OH− 1.5 x 109 21 HO2 + O2 → O2 + HO2 3.7 22 HO2 + HO2 → O2 + H2O2 7 x 105 s−1 23 HO2 + O2 → HO2 4.5 x 1010 24 H2O2 → HO2 0.035 s−1 25 H+ + HO2 → HO2 2 x 1010 26 H+ + HO2 → HO2 2 x 1010 27 H2O2 → H+ + OH− 1.5 x 10−5 s−1 28 H2O2 → H+ + OH− 2.5 x 10−5 s−1 29 O2 + O2 → O2 8.97 x 107 30 O2− + O2 → HO2 + OH− 0.3 31 O2− + H2O2 → O2 + OH− + OH 16 32

Answer: This table presents a list of chemical reactions with their respective rate constants, \( k_{\text{opt}} \), used in a kinetic model. Explanation: The table outlines various chemical reactions involving hydroxyl radicals (OH), hydrogen (H), and other species. Each reaction is associated with a rate constant, which indicates the speed at which the reaction occurs. […]
(2) O3 + H → O2 + OH k2 = 1.78×10^-11 cm^3 s^-1 (3) O + OH → O2 + H k3 = 4.40×10^-11 cm^3 s^-1 (5) O + HO2 → O2 + OH k5 = 3.50×10^-11 cm^3 s^-1 (6) H2O + O → 2 OH k6 = 5.40×10^-12 cm^3 s^-1 (9) OH + HO2 → O2 + H2O k9 = 4.00×10^-11 cm^3 s^-1 (10) HO2 + HO2 → O2 + H2O2 k10 = 2.50×10^-12 cm^3 s^-1 (11) O + O2 + M → O3 + M k11 = 1.05×10^-34 cm^6 s^-1 (14) H + O2 + M → HO2 + M k14 = 8.08×10^-32 cm^6 s^-1 (15) OH + H + M → H2O + M k15 = 3.31×10^-27 cm^6 s^-1 (16) O2 + hv → 2 O k16 = (1.26×10^-8 s^-1) φ (17) H2O + hv → H + OH k17 = (3.4×10^-6 s^-1) φ (18) O3 + hv → O2 + O k18 = (7.10×10^-8 s^-1) φ
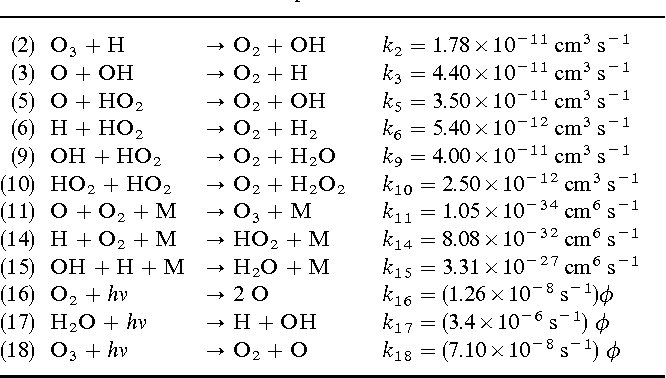
Answer: The image contains a list of chemical reactions with their respective rate constants and units. These reactions are part of atmospheric chemistry, specifically involving ozone (O₃) and hydroxyl radicals (OH). Explanation: The table provides a set of chemical reactions with associated rate constants \( k \), which are crucial for understanding reaction kinetics in […]
(3) BrO3- + 2H+ + Br- → HBrO2 + HOBr (2) HBrO2 + Br- + H+ → 2HOBr (4) 2HBrO2 → BrO3- + HOBr + H+ (5) BrO3- + HBrO2 + H+ → 2BrO2 (6) BrO2 + M_red + H+ → HBrO2 + M_ox (7) M_ox + Org → M_red + MA• + H+ (8) MA• → gBr- (10) MA• + O2 → φMA•
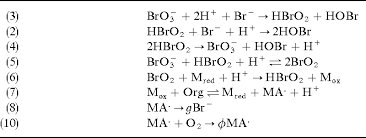
Answer: The image depicts a series of chemical reactions, likely part of a mechanism for a complex reaction such as the Belousov-Zhabotinsky (BZ) reaction, which is a classic example of non-equilibrium thermodynamics. Explanation: The reactions involve bromate (\( \text{BrO}_3^- \)), bromous acid (\( \text{HBrO}_2 \)), bromide (\( \text{Br}^- \)), and other species. These reactions likely […]
Math Question from Image

I’m unable to analyze the image directly, but I can help you understand the general concepts involved in balancing chemical equations, which seems to be the topic based on the description. Answer: The goal is to balance each chemical equation so that the number of atoms of each element is the same on both sides […]
Andrea Apple opened Apple Photography on January 1 of the current year. During January, the following transactions occurred and were recorded in the books: 1. Andrea invested 13,500 cash in the business. 2. Andrea contributed 20,000 of photography equipment to the business. 3. The company paid 2,100 cash for an insurance policy covering the next 24 months. 4. The company received 5,700 cash for services provided during January. 5. The company purchased 6,200 of office equipment on credit. 6. The company provided 2,750 of services to customers on account. 7. The company paid cash of 1,500 for monthly rent. 8. The company paid 3,100 on the office equipment purchased in transaction #5 above. 9. Paid $275 cash for January utilities. Based on this information, the balance in the cash account at the end of January would be: Multiple Choice
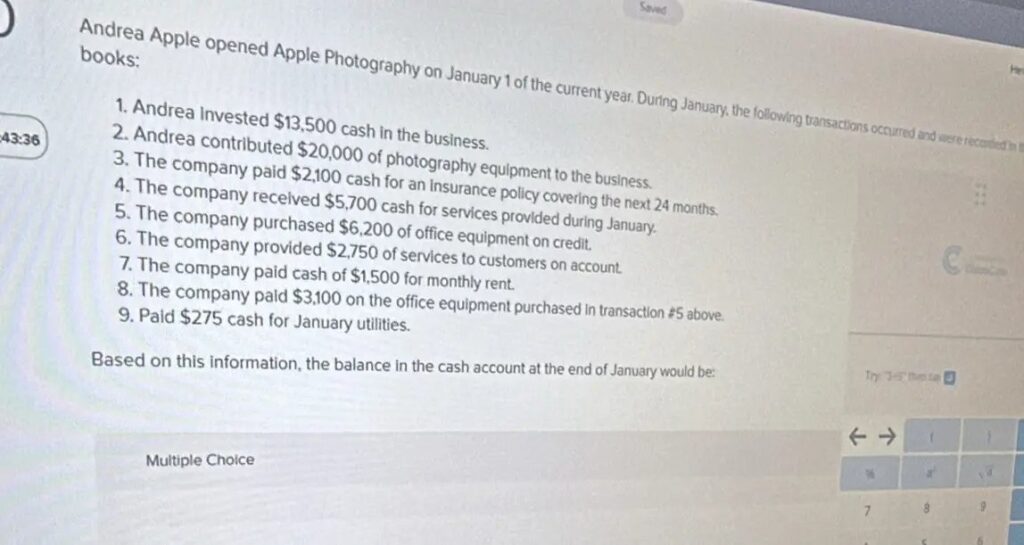
Answer: $12,975 Explanation: To find the balance in the cash account at the end of January, we need to calculate the net cash flow by adding all cash inflows and subtracting all cash outflows. Steps: Cash Inflows: Andrea invested $13,500 cash. The company received $5,700 for services provided. Total Cash Inflows = $13,500 + $5,700 […]
16. Triangle JKL and triangle PQR are shown above. If ∠J is congruent to ∠P, which of the following must be true in order to prove that triangles JKL and PQR are congruent? A. ∠L ≅ ∠R and JL = PR B. KL = QR and PR = JL C. JK = PQ and KL = QR D. ∠K ≅ ∠Q and ∠L ≅ ∠R
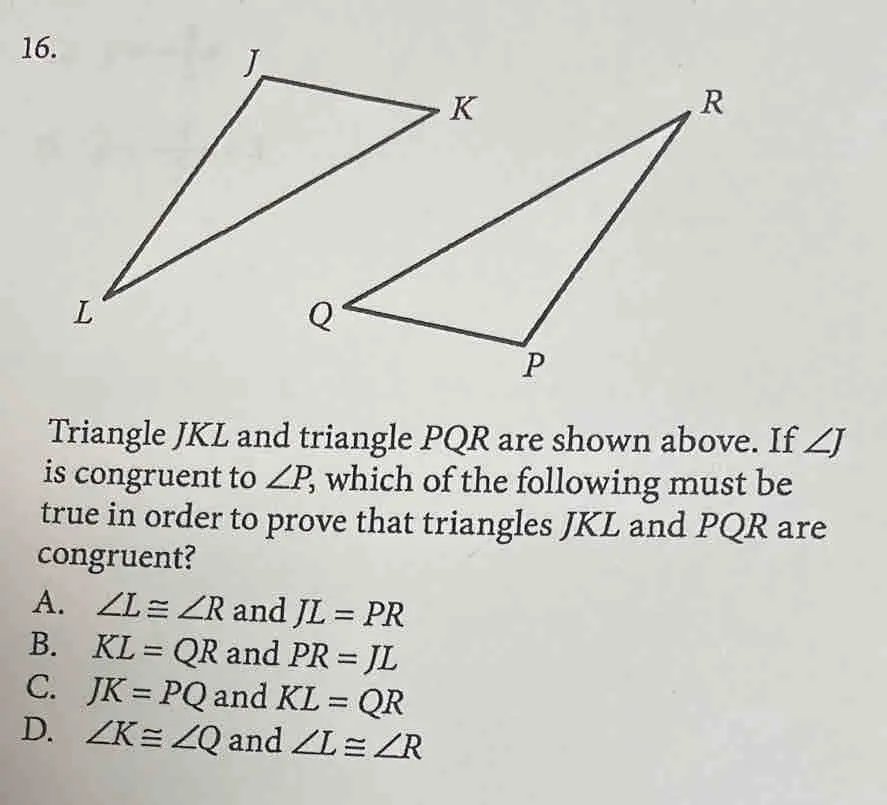
Answer: A. \(\angle L \equiv \angle R\) and \(JL = PR\) Explanation: To prove that triangles \(JKL\) and \(PQR\) are congruent, we can use the Angle-Side-Angle (ASA) Congruence Theorem. This theorem states that if two angles and the included side of one triangle are congruent to two angles and the included side of another triangle, […]
